Have you ever wondered if something is acidic or basic? Many people search this question when studying chemistry, cooking, health, or water quality. Words like acid, base, and pH level appear in school books, lab reports, and even food labels. But the meaning can feel confusing.
For example, lemon juice tastes sour, soap feels slippery, and baking soda is used in cooking. Are these acidic or basic? People also ask questions like “What is acidic and basic?”, “What pH level is acidic basic?”, and “What is an acid base?”. Students often look for acidic basic notes, acidic basic notes pdf, or a simple acidic basic formula to understand the topic better.
This guide explains everything in simple English. You will learn the acid and base definition and examples, how the pH scale works, and how to identify substances that are acidic, basic, or neutral. We will also show real-life acidic basic examples and explain how the terms are used in school, science, and daily life.
By the end, you will clearly understand what acid and base mean and how to tell if something is acidic or basic.
Acidic or Basic –Meanings
The quick answer is simple.
A substance is acidic if it has a pH level below 7.
A substance is basic (alkaline) if it has a pH level above 7.
A substance is neutral if the pH is exactly 7.
Examples
| Substance | Type | pH Level |
|---|---|---|
| Lemon Juice | Acidic | ~2 |
| Vinegar | Acidic | ~3 |
| Pure Water | Neutral | 7 |
| Baking Soda | Basic | ~8 |
| Soap | Basic | ~10 |
So if someone asks “What is acidic and basic?”, the answer is about pH level and chemical behavior.
Acidic substances release hydrogen ions (H⁺).
Basic substances release hydroxide ions (OH⁻).
This is the core acidic basic formula used in chemistry.
The Origin of Acidic or Basic
The word acid comes from the Latin word “acidus”, which means sour. This makes sense because many acids taste sour. For example:
- Lemon
- Vinegar
- Orange juice
The word base was introduced by the chemist Justus von Liebig and later expanded by scientists like Svante Arrhenius and Johannes Brønsted. These scientists developed the modern acid and base definition and examples used in chemistry today.
Key Scientific Definitions
Arrhenius Definition
- Acid → Produces H⁺ ions in water
- Base → Produces OH⁻ ions in water
Brønsted–Lowry Definition
- Acid → Proton donor
- Base → Proton acceptor
These definitions appear in many acidic basic notes pdf used in schools and universities.
British English vs American English Spelling
Unlike many English words, acidic and basic spelling is mostly the same in both British and American English. However, the related word alkaline is often used more in British contexts.
Comparison Table
| Term | American English | British English | Meaning |
|---|---|---|---|
| Acidic | Acidic | Acidic | pH below 7 |
| Basic | Basic | Basic | pH above 7 |
| Alkaline | Used sometimes | Used more often | Another word for basic |
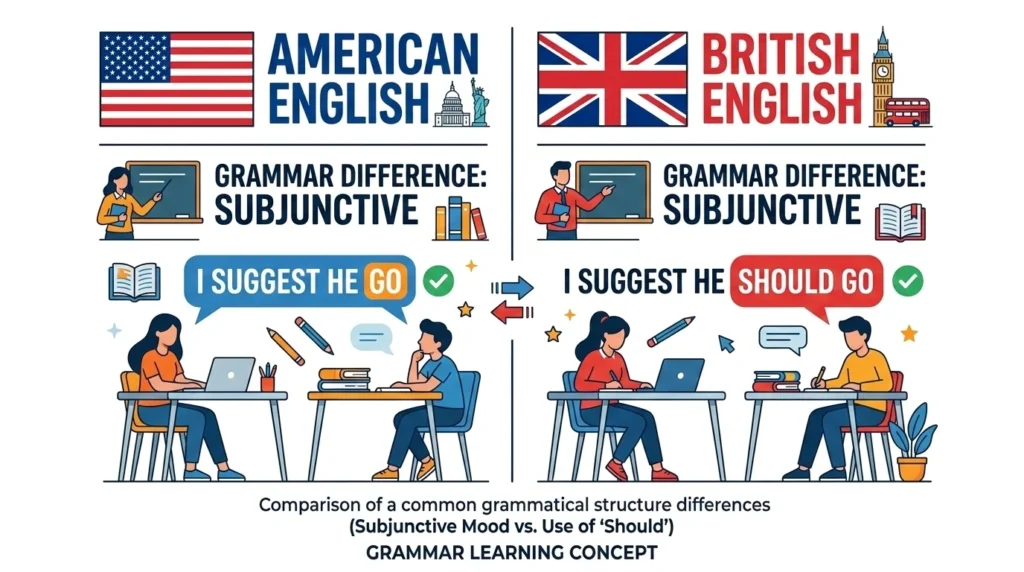
Example:
US style:
“The solution is basic.”
UK style:
“The solution is alkaline.”
Both mean the same thing.
Which Spelling Should You Use?
Your choice depends on your audience.
If your audience is in the United States
Use acidic and basic.
This is the most common style in American textbooks.
If your audience is in the UK or Commonwealth countries
You can use acidic and alkaline more often.
For global or online writing
Use acidic and basic.
This is the clearest and most widely understood wording in science and education.
Common Mistakes with Acidic or Basic
Many students make small mistakes when learning about acids and bases.
Mistake 1: Confusing Acid with Strong Acid
Not all acids are strong.
Example:
- Lemon juice → weak acid
- Hydrochloric acid → strong acid
Mistake 2: Thinking Basic Means Safe
Some bases are very dangerous.
Example:
- Sodium hydroxide (drain cleaner) is extremely strong.
Mistake 3: Forgetting Neutral Substances
People often ask “What is acidic basic and neutral?”
Remember:
- Acidic → pH < 7
- Neutral → pH = 7
- Basic → pH > 7
Mistake 4: pH Scale Confusion
Many people search “What pH level is acidic basic?”
Correct rule:
| pH Range | Type |
|---|---|
| 0–6 | Acidic |
| 7 | Neutral |
| 8–14 | Basic |
Acidic or Basic in Everyday Examples
Understanding acids and bases is useful in daily life.
In Food
- Lemon juice → acidic
- Coffee → slightly acidic
- Baking soda → basic
In Cleaning
Soap and detergents are basic.
They help break down oils and grease.
In Health
Your stomach contains hydrochloric acid to digest food.
In Agriculture
Farmers test soil to see if it is acidic or basic before planting crops.
Example in Writing
Email example:
“The water test shows the solution is slightly acidic with a pH of 6.”
News example:
“Scientists found the ocean is becoming more acidic due to climate change.”
Social media example:
“Fun fact: Baking soda is basic, but lemon juice is acidic!”
Acidic or Basic – Google Trends & Usage Data
Search data shows that many students and researchers look for topics like:
- Acidic basic notes
- Acidic basic pdf
- Acidic basic formula
- Acidic basic examples
These searches are common in countries with large student populations.
High Search Countries
- India
- Pakistan
- United States
- United Kingdom
- Canada
Students often search for acid and base definition and examples while studying chemistry.
The term “what is acid and base” is also common among beginners learning science.
Acidic vs Basic Comparison Table
| Feature | Acidic | Basic |
|---|---|---|
| pH Level | Below 7 | Above 7 |
| Taste | Sour | Bitter |
| Ion Produced | H⁺ | OH⁻ |
| Examples | Lemon, vinegar | Soap, baking soda |
| Litmus Test | Turns blue litmus red | Turns red litmus blue |
These are the most common acidic basic examples taught in science.
FAQs
What is acidic and basic?
Acidic substances have a pH below 7, while basic substances have a pH above 7. Neutral substances have pH 7.
What is acid and base?
An acid releases hydrogen ions (H⁺) in water.
A base releases hydroxide ions (OH⁻) or accepts protons.
What pH level is acidic basic?
- Acidic → pH 0–6
- Neutral → pH 7
- Basic → pH 8–14
What is an acid base reaction?
An acid-base reaction occurs when an acid reacts with a base to form salt and water.
Example:
HCl + NaOH → NaCl + H₂O
What is acidic basic and neutral?
These terms describe the pH level of a substance:
- Acidic → below 7
- Neutral → 7
- Basic → above 7
Where can I find acidic basic notes pdf?
Students usually find acidic basic notes pdf in:
- Chemistry textbooks
- Educational websites
- School learning platforms
What are simple acidic basic examples?
Examples include:
Acidic:
- Lemon juice
- Vinegar
Basic:
- Soap
- Baking soda
Neutral:
- Pure water
Conclusion
Understanding whether something is acidic or basic is an important part of chemistry and daily life. The key idea is the pH scale, which helps us measure how acidic or basic a substance is. If the pH is below 7, the substance is acidic. If the pH is above 7, it is basic. A pH of 7 means the substance is neutral.
Acids usually taste sour and produce hydrogen ions, while bases often feel slippery and produce hydroxide ions. Common acidic basic examples include lemon juice, vinegar, baking soda, and soap. These examples help students understand the topic more easily.
Many learners search for acidic basic notes, acidic basic pdf, or the acidic basic formula to study chemistry concepts. Knowing the acid and base definition and examples helps in science education, laboratory work, and even everyday activities like cooking and cleaning.
In simple terms, learning what acid and base mean helps us understand how substances react, how food behaves, and how many natural processes work.
Once you understand the pH scale and basic definitions, identifying whether something is acidic, basic, or neutral becomes very easy.

“Elizabeth Gaskell was a Victorian novelist known for compassionate social realism, memorable characters, and classics like North and South and Cranford.”